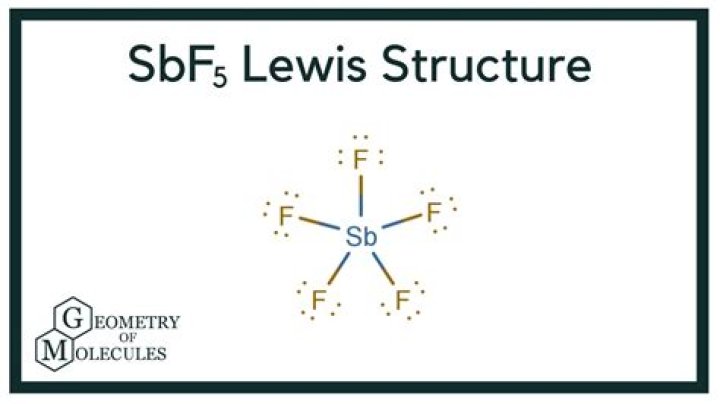
SbF5 forms a trigonal bipyramidal shape.
What is the bond angle of SbF5?
The bond angles for SbF5 are 180° and 90°.
What is the hybridization of if5?
Since the central atom iodine has 7 valence electrons out of which 5 electrons form 5 sigma bonds with F atoms and and 2 electrons form 1 lone pair making the stearic number 6 which implies the hybridization of the central atom is sp3d2 where the geometry is octahedral and the shape is square pyramidal.
What shape is if4?
So Hybridization = [s{p^3}{d^2}], the shape of the molecule is octahedral geometry, but the geometry of the atom takes a square planar shape is due to iodine carries two lone pairs of electrons, one above the plane and one below the plane hence the shape of the molecule is square planar.
Does SbF5 have dipole dipole forces?
Since there are four such axis in the molecule, it cannot have a dipole moment. Note however, that each of the bonds is certainly polar. They just cancel out, as could be proven by vectorial addition of the bond dipole moment. The bond dipole moments in vertical axis cancel out .
Are BrF5 bonds polar?
Because of its square pyramidal molecular structure, asymmetric charge distribution, and 90° bong angle, bromine pentafluoride (BrF5) is a polar molecule.
What is the hybridization of SF6?
The hybridization of SF6 is sp3d2 type. Just to describe the compound in brief, Sulphur Hexafluoride is a type of greenhouse gas which is colourless, odourless, non-toxic and non-flammable.
What is the shape of IF5?
Since it has 5 bond pair and one lone pair hence it shows square pyramidal geometry instead of octahedral geometry.
What is the shape of I5?
The anion I5- is bent, with a 95 angle at the central iodine atom.
What is the hybridization of I3?
I3- ion is basically sp3d hybridized.
Is SbF5 trigonal bipyramidal?
In the gas phase, SbF5 adopts a trigonal bipyramidal structure of D3h point group symmetry (see picture). The material adopts a more complicated structure in the liquid and solid states.
Is CS2 a polar covalent bond?
Although the electronegativity of Carbon(2.55) and Sulfur(2.58) differs slightly, making the C-S bonds are slightly polar, the molecule is nonpolar due to the symmetric linear form of the CS2 molecule. Both C-S bonds have equal and opposite dipoles that cancel each other out, making the CS2 molecule non-polar.
What is the polarity of iodine?
Iodine is soluble, as iodine is a non-polar molecule. The iodine molecules and cyclohexane molecules form weak intermolecular attractions. Valence electrons of Mg atoms are free to move throughout the structure.