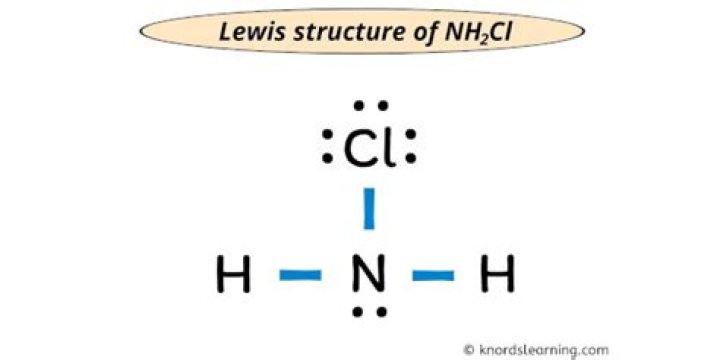
The molecular geometry of NH2Cl is trigonal pyramidal and its electron geometry is tetrahedral. The lewis structure of NH2Cl has 6 bonding electrons and 8 nonbonding electrons.
Is nh2cl2 polar or nonpolar?
Decision: The molecular geometry of NH2Cl is trigonal pyramidal with asymmetric charge distribution on the central atom. Therefore this molecule is polar.
How many valence electrons does nhcl2?
Drawing the Lewis Structure for NH2Cl
Viewing Notes: There are a total of 14 valence electrons in the NH2Cl Lewis structure. Nitrogen (N) is the least electronegative atom and goes at the center of the NH2Cl Lewis structure. Hydrogen atoms always go on the outside of a Lewis structure.
What intermolecular forces are present in nh2cl?
NH2Cl is also a polar molecule, so there will be dipole-dipole interaction. And as with any molecule, there will be dispersion forces.
What is the central atom of the molecule nh2cl?
it has a central nitrogen atom and the other atoms are terminal.
Is BeBr2 polar or nonpolar?
The center beryllium atom of BeBr2 has no lone pairs of electrons, resulting in linear electron geometry. However, the molecular geometry of BeBr2 is linear in nature. It’s the BeBr2 molecule’s symmetrical geometry. As a result, the BeBr2 molecule is nonpolar.
What is the molecular shape of Hof?
The compound has been characterized in the solid phase by X-ray crystallography as a bent molecule with an angle of 101°.
What makes a trigonal pyramidal?
Trigonal pyramidal geometry is also shown by molecules having four atoms or ligands. Central atom will at the apex and three other atoms or ligands will be at one base, where they are in the three corners of a triangle. There is one lone pair of electrons in the central atom.
What is the point group of CH2Cl2?
CH2Cl2 1)belongs to point group D2 2)has one plane of symmetry, sigma h 3)has one C2 axis of rotation | Study.com.
What shape is NF3?
The molecular geometry or shape of NF3 is a trigonal pyramid and its electron geometry is tetrahedral. NF3 lewis dot structure contains 1 lone pair and 3 bonded pairs.
What kind of intermolecular forces act between two water molecules?
Water molecules are held together by hydrogen bonds. Hydrogen bonds are a much stronger type of intermolecular force than those found in many other substances, and this affects the properties of water.
What types of intermolecular forces are expected between ch3ch2ch2ch2oh molecules?
The molecules of 1-butanol experience three types of intermolecular forces namely, London dispersion forces, dipole-dipole interactions, and hydrogen See full answer below.
What type s of intermolecular forces are expected between ch3ch2ch2ch3 molecules?
Therefore, the intermolecular forces also include dipole forces.